Monogram Orthopedics takes a personalized approach to joint replacement. The company's robotic arm prepares the bone for ...
There may be other available opportunities that are similar to this investment but have different attributes, characteristics, cost factors, and fees.
Please review company financials carefully.
Disclosures & Disclaimers
Problem
Robotic joint replacement isn't where it needs to be
A leader has emerged, but market adoption is lagging. The industry is waiting for a disruptive solution.
We all know someone with a joint replacement – and it can be a scary surgery. Almost 100,000 TKA (total knee arthroplasty) procedures fail annually, and only 66% of patients felt their knee felt “normal” after the procedure. Over half of knee replacement patients experienced residual symptoms.
Over the past few years, only one major system has dominated the robotic joint replacement market (approximately 90% of orthopedic robotic surgeries). However, robot utilization remains low (88% of knee replacements are still manual) for a number of reasons:
- Speed
Manual surgery can be relatively faster. Nearly every robotics limitation impacts speed — and, by extension, surgeon profitability. Current robotic solutions tend to result in higher cost by $3K per case. - Clinical benefits are mixed
Early-market products have been less effective than expected. - High fiddle factor
Technical limitations require extensive adjustments and workarounds. - Underwhelming first impression
Even market-leading products feel like an incremental improvement, and not truly transformative.
Our vision is to facilitate the next generation of 3D printed personalized implants and to make surgical robotics ubiquitous over the next decade.
ONN, 2021 Hip and Knee Implant Review (Volume 33, Number 3, August 2022)
Nam, D., Nunley, R. M., & Barrack, R. L. (2014). Patient dissatisfaction following total knee replacement. The Bone & Joint Journal, 96-B(11_Supple_A), 96–100.
Solution
Monogram is ready

We've identified and tried to address major opportunities for improvements to the the surgical workflow. Our focus is speed with uncompromised accuracy, reducing the average procedure time from 70 minutes to 6 minutes.
We're combining 3D-printed implants personalized for each patient with robotically-precise surgery, creating a more stable knee replacement that won't comprise a patient's health.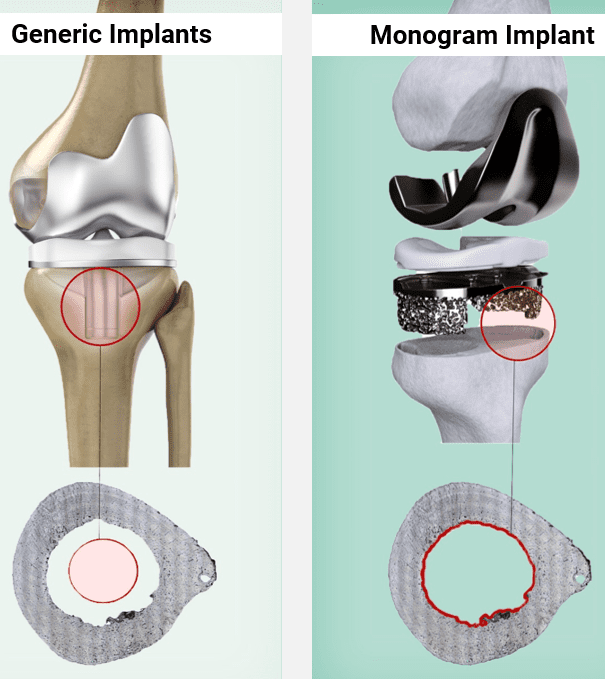
While cemented fixation is currently the gold standard, it is a somewhat outdated method. Cementless fixation relies on porous surfaces to act as substrates for bone ingrowth to provide a biological bond that continually remodels over time. The benefits are two-fold: 1) less loosening and 2) more efficient surgery.
Monogram has designed a tibial tray prototype with a low-height peripheral fin that relies on engagement with the most supportive available bone vs. current implants that rely on the soft central bone for fixation.
Press-fit future The new standard of orthopaedic implants will be driven by robotics |
|---|
|
Product
Proven keys
to robotic success
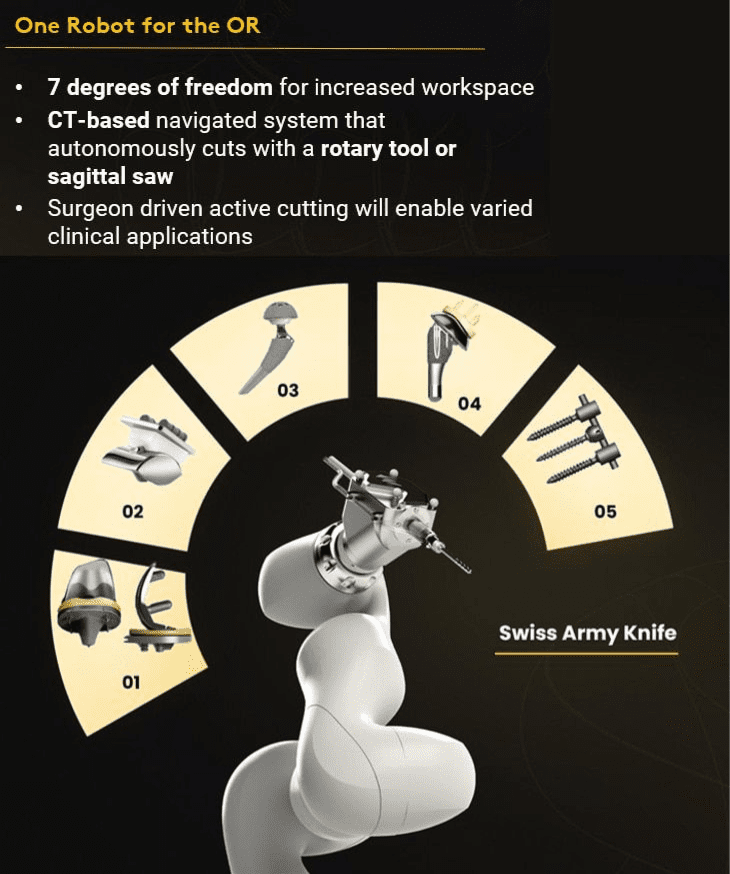
Monogram has designed its robotic platform to be highly scalable — the vision for the future is to have one robot for the operating room. We also anticipate that implants in the future will be increasingly personalized. Robots need to enable this future. Some of the features of the Monogram robot include:
- CT-based planning
Monogram utilizes advanced imaging to plan patient-specific surgeries for improved accuracy and virtualization.
Navigated
Our system tracks the real-time patient position and the real-time robot position for safety- Multiple cutting modalities
The Monogram robot will be designed to cut with a sagittal saw or with a rotary tool. A sagittal saw for speed and efficiency and a rotary tool for shaping bone.
We've done over 50 simulated surgeries in our cadaver lab, including surgeries with our surgeon panel to solicit feedback. We think our solution has the potential to advance the state-of-the-art in orthopedic robotics. Watch the Monogram robot complete registration and the 6 TKA cuts in a simulated cadaveric surgery in our lab!
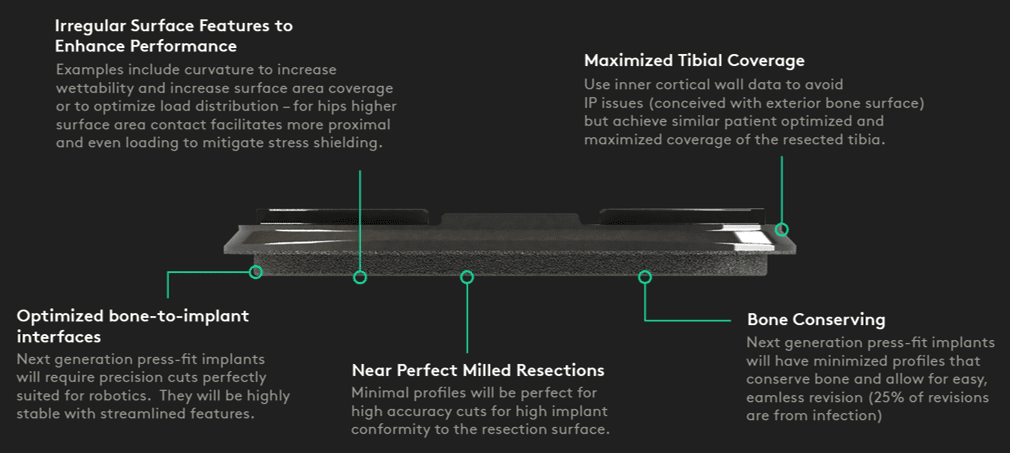
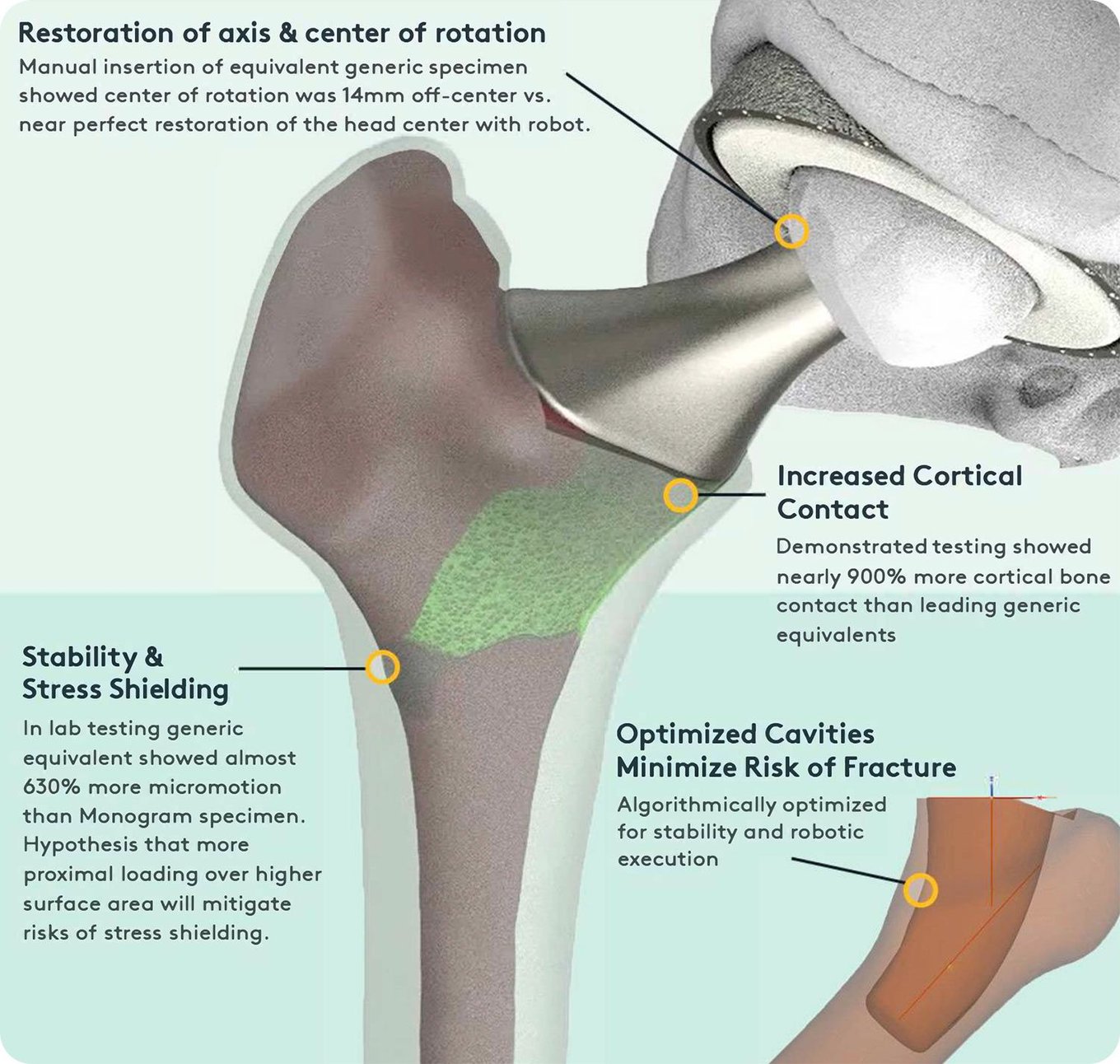
Monogram believes the future of orthopedics lies in the combination of robotics and 3D-printed patient-optimized implants. Monogram has novel knee and hip designs that have patents that have been filed. The main design objective is to improve stability in a bone-conserving form factor. The Monogram 3D-printed implants will be designed to be:
Bone-conserving
By leveraging preoperative patient data, we believe can design implants that conserve bone.Highly stable
Our research with UCLA and UNMC indicates that the Monogram implants are highly stable
Gen 2 | Novel Monogram 3D-printed tibial component + press-fit tibia

Traction
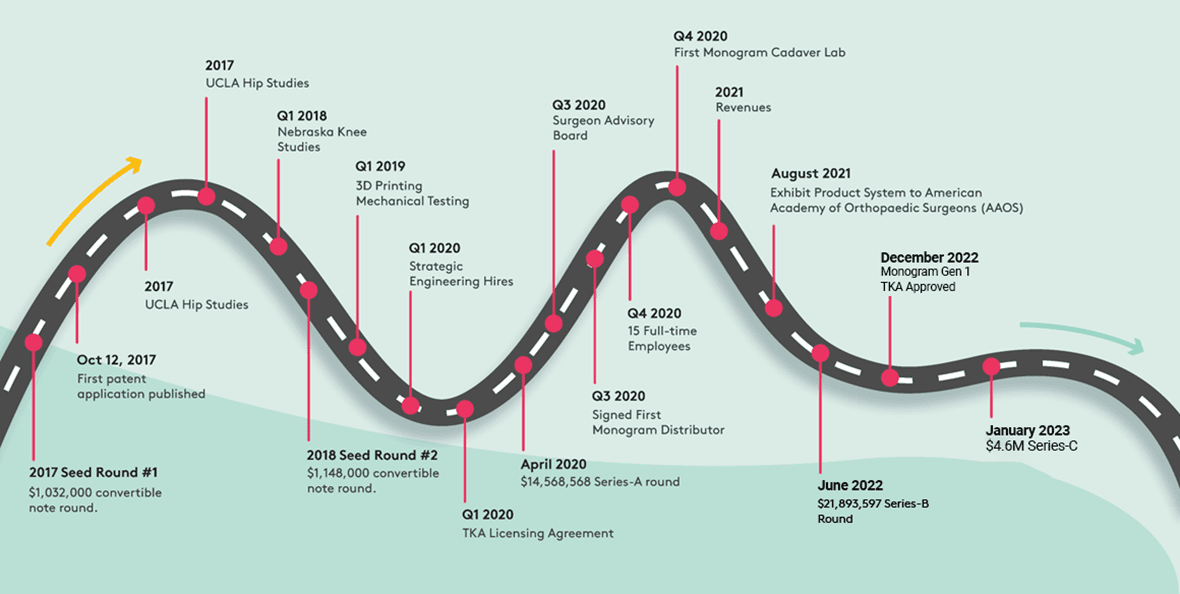
The Monogram roadmap
The most important milestone in the coming 2 months is the planned public listing on NASDAQ immediately following the close of this offering.
Historical Milestones
- 20 patent filings (status pending) for robotics and novel implants
- National Science Foundation (NSF) grant award
- FDA-approved TKA (licensed), UKA and THA implants (licensed)*
- Over 50 simulated surgeries performed in Monogram's cadaver lab
- Achieve 6-min registration and TKA bone prep with prototype in cadaver lab
Upcoming milestones
- FDA pre-submission meeting (completed Q1 2023, supplement Q2)
- Public listing on Nasdaq (expected Q2 2023)
- Partial press-fit knee FDA submission (expected 2023)
- Expected sales in 2024 (assumes favorable FDA meeting)
Average time to approval per FDA: <4 months
Our development efforts are only accelerating. The most critical objective for our company is to obtain FDA approval for our technology as quickly as possible.
*UKA: Unicompartmental (partial) Knee Arthroplasty; THA: Total Hip Arthroplasty
Customers
Monogram's beneficiaries
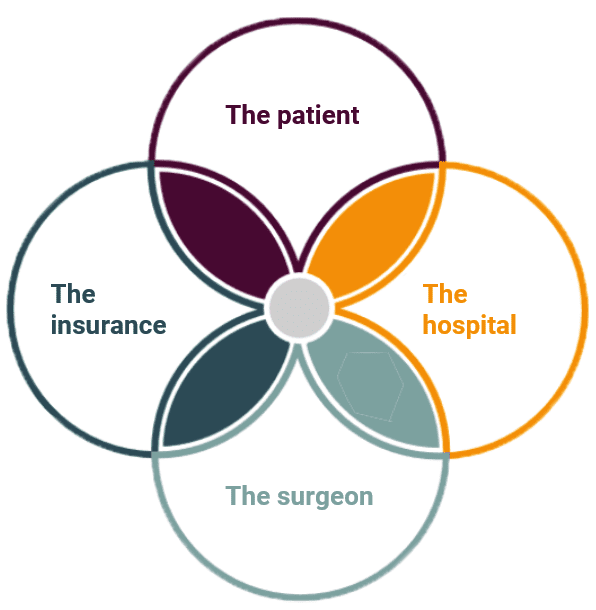
Monogram has four major beneficiaries from which it must secure buy-in to be successful, specifically:
The Patient
Monogram exists to serve the interests of patients. The safety and well-being of the patient are paramount. The thing most patients care about is maximizing the probability of a favorable outcome.The Surgeon
The surgeons are the product champions – to place systems in hospitals requires surgeons that advocate for your product to hospital purchasing departments. They generally aren't willing to compromise volume with technology that might slow them down. To identify surgeon advocates, Monogram has made sure to involve its surgeon panel in its development efforts. Monogram will leverage its cadaver lab to give surgeons the opportunity to demo the system. Initial user feedback has exceeded expectations!- The Hospitals
Hospitals' primary interest is to mitigate readmissions and complications and to do so without introducing excessing incremental costs. With elective surgery like knee replacements, hospitals must aggressively compete for patients. Technology can be useful for attracting patient interest. A lot of hospitals don't have un-budgeted capital for an expensive surgical system. If needed, we will aggressively discount robots because, ultimately, our interest is in selling implants. They also don't want to compromise the volume they can do with slow technology.
The Payer
Insurance companies care about normalizing patient outcomes and mitigating risk. If a technology mitigates risk and it's supported by clinical data, they are generally supported. We should note that robotic TKA has well-established insurance billing codes, which is helpful. We aren't inventing a new surgery.
In the development of its surgical robot, Monogram has carefully considered every major benefactor.
Business model
Our revenue model
Orthopedic robotics is a textbook "razor-razorblade model." The goal is to place robots (even at or below cost) because the lion's share of the profits is in the utilization of the robot and the sale of implants. Monogram believes it could even give the robots away if necessary to penetrate the market in the early stages.

Market
Significant growth in robotic procedures by 2027
Monogram is developing differentiated products for some of the fastest growing segments of the market
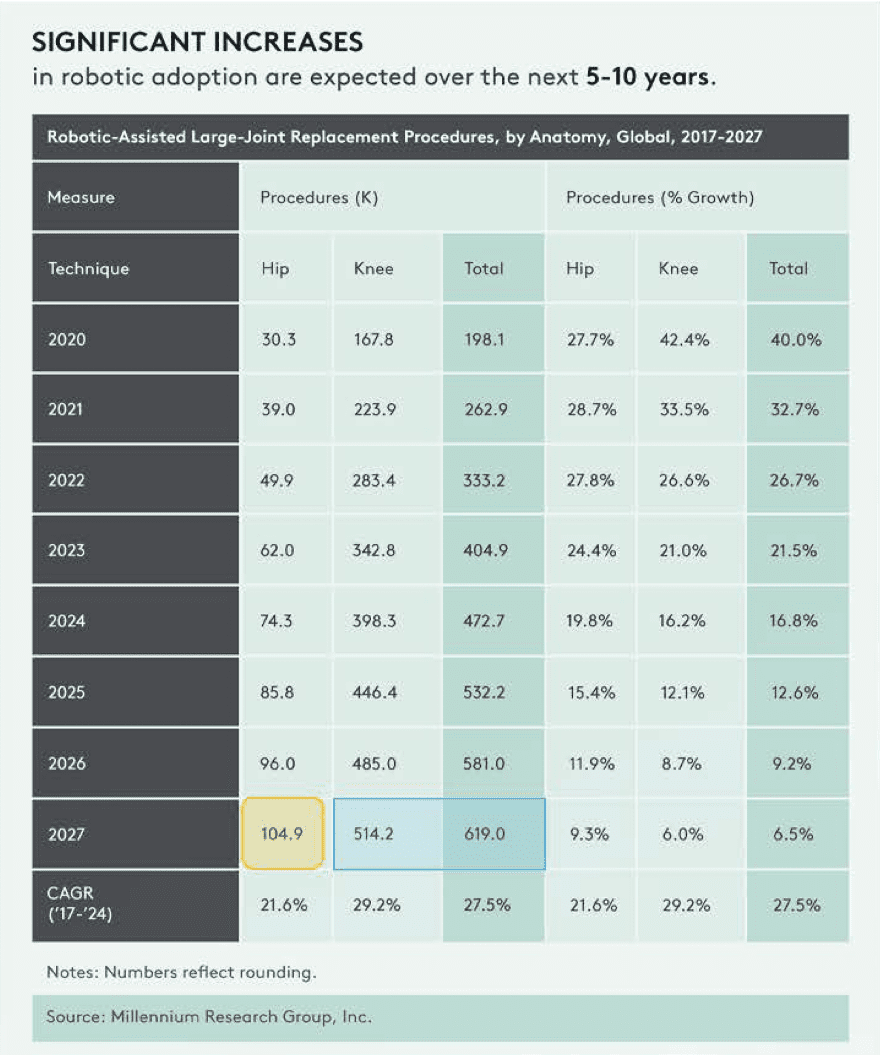
Despite the considerable market size, only 8% of total knee replacement procedures today are robotic, 3% of hips, and 3% of shoulders. Monogram's first target market (robotic TKA) could double in the next three years.
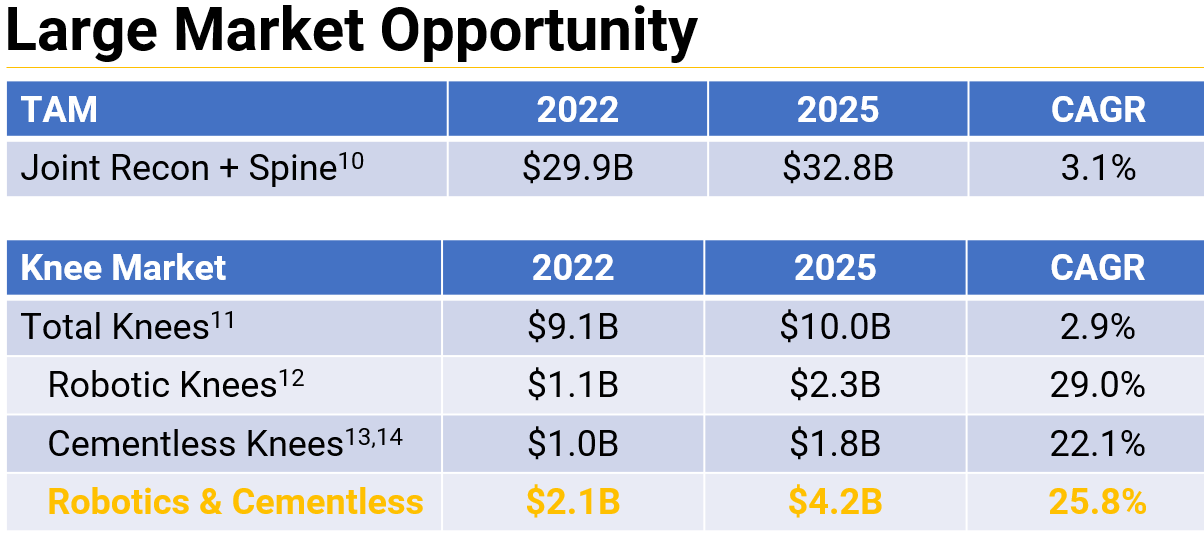
The press-fit knee market is also large and growing. We expect a rapid change in mix to cementless. Monogram also anticipates that clinical data will help drive robotic adoption. Clinical data suggests that the 90-day global expenditures could be $4,049 less for robotic TKA vs. Manual TKA, and in addition, a transition to functional/kinematic alignment could be a robotic driver.
Competition
The market is highly consolidated with the top four players accounting for over 80% of the knee market. Stryker (Mako) accounts for approximately 90% of robotic joint replacement surgeries. As far as we are aware, of the big four, Mako is the only CT-based robot and the only arm based robot with a robot mounted sagittal saw.
On the implant side, the differentiation is not as clear. Generally speaking, all implants look generic and geometric. Monogram anticipates that a novel robotic system that enables the next generation of 3D printed press-fit implants will also differentiate the implants.
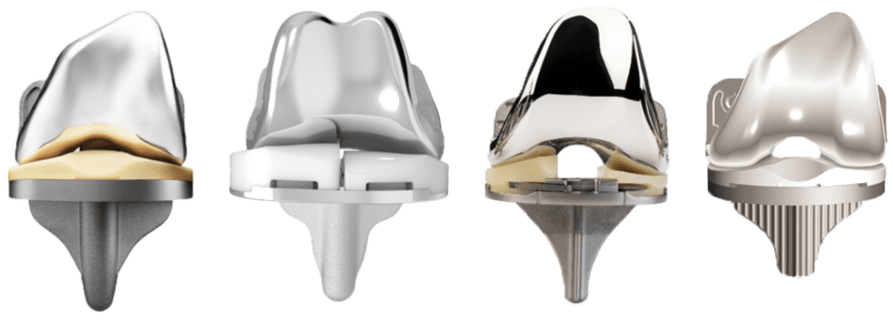 Knee implants
Knee implants
Monogram will compete with advanced robotics combined with novel implants.
Stryker (Mako) has set the standard for speed and efficiency in orthopedic robotics. Monogram's focus is to develop a surgical robot that is time-competitive with the Mako system.
Vision and strategy
Clear roadmap for success
Monogram aims to follow the roadmap that was established by Mako Surgical (acquired by Stryker).
Although we cannot guarantee, Monogram's goal will be to increase sales dramatically within 5-years of FDA approval.
Impact
Improving the standard of care in orthopedics
Monogram's mission is to try and push forward the standard of care in orthopedics with technological advances. Only 66% of patients felt their knee felt “normal,” with 54% experiencing residual symptoms. Approximately 100,000 knees fail annually. Our vision is to make robots ubiquitous within the next 10 years. We think Monogram's vision is an extremely high impact, and it's why so many of our talented engineers choose to support our mission.
The video below demonstrates some of the challenges we are trying to address with our technology.
Younger (<65 years) and obese patients require fixation less prone to mechanical loosening over time. The higher demands associated with these patients can overload the implants and increases risk for aseptic loosening. Aseptic, mechanical loosening is the now most common reason for late (>2y) revision surgery, accounting for 40% of cases, especially for these higher demand patients. The tibial component is >12 times more likely to fail compared to the femoral component. These trends and sheer surgical volume will contribute to a revision burden projected to increase by up to 182% by 2030, with costs averaging over $49K per revision surgery. Mechanical loosening is the now most common reason for late (>2y) revision surgery.
Funding
Over $38M raised
from more than 17,000 investors

Leadership
Our leadership team
Strong surgeon support
Medical advisory board

Summary
Monogram is addressing the $29B orthopedic market, starting with the $9B knee market (786,000 total knee replacements in the US per year) with novel robotics that will eventually enable the future of 3D printed press-fit personalized implants.
- Planned public listing on NASDAQ (reserved ticker: MGRM)
- 20 patent filings (status pending) – one of the first active navigated sagittal saw mounted robot
- High-performance surgical robotic prototype
- FDA-approved press-fit TKA implant, 3D printed next-gen pipeline (licensed)
- Over 50 simulated cadaveric surgeries – 6-min registration & bone cutting
- 26 FTE employees
Disclaimers
This page may include ''forward-looking statements.'' To the extent that the information presented in this presentation discusses financial projections, information, or expectations about Monogram Orthopedics Inc.’s business plans, results of operations, products or markets, or otherwise makes statements about future events, such statements are forward-looking. Such forward-looking statements can be identified by the use of words such as ''should,'' ''may,'' ''intends,'' ''anticipates,'' ''believes,'' ''estimates,'' ''projects,'' ''forecasts,'' ''expects,'' ''plans,'' and ''proposes.'' Although Monogram Orthopedics Inc. believes that the expectations reflected in these forward-looking statements are based on reasonable assumptions, there are a number of risks and uncertainties that could cause actual results to differ materially from such forward-looking statements. You are urged to carefully review and consider any cautionary statements and other disclosures, including the statements made under the heading "Risk Factors" and elsewhere in the offering statement filed with the SEC. Forward-looking statements speak only as of the date of the document in which they are contained, and Monogram Orthopedics Inc. does not undertake any duty to update any forward-looking statements except as may be required by law.
The offering will be made only by means of an offering circular. An offering statement on Form 1-A relating to these securities has been filed with the U.S. Securities and Exchange Commission and has become qualified. The securities offered by Monogram are highly speculative. Investing in shares of Monogram involves significant risks. The investment is suitable only for persons who can afford to lose their entire investment. Furthermore, investors must understand that such investment could be illiquid for an indefinite period of time. No public market currently exists for the securities, and if a public market develops following the offering, it may not continue.
Monogram intends to list its securities on a national exchange and doing so entails significant ongoing corporate obligations including but not limited to disclosure, filing and notification requirements, as well compliance with applicable continued quantitative and qualitative listing standards. For additional information on Monogram, the offering and any other related topics, please review the Form 1-A offering circular that can be found at the following location EDGAR Entity Landing Page (sec.gov). Additional information concerning Risk Factors related to the offering, including those related to the business, government regulations, intellectual property and the offering in general, can be found in the risk factor section of the Form 1-A offering circular.
Risks of early stage investment. Not an offer to buy or sell securities. This is a long-term speculative illiquid investment. Investment is not FDIC or SiPC insured. You may lose money.
Your investment is binding and irrevocable, although we reserve the right to reject it for any reason or no reason at all. Funds committed will remain in an escrow account maintained by BankProv until such time as a closing occurs. Securities offered through OpenDeal Broker LLC, a registered broker dealer, member of FINRA (www.finra.org), member of SIPC (www.sipc.org). We will pay OpenDeal Broker LLC, a registered broker-dealer, a cash commission equal to 7% of the amount raised through the Republic Platform, plus certain offering costs. Please review OpenDeal Broker LLC’s Form CRS. OpenDeal Broker LLC may require additional documents or information from you to complete your purchase, you will be contacted by a registered representative in this event.
An offering statement relating to Monogram Orthopedics Inc.’s Common Stock has been filed with the Securities and Exchange Commission and became qualified on March 1, 2023. Prior to making any investment in Monogram Orthopedics Inc.’s Common Stock, you should review a copy of the offering circular or contact Monogram Orthopedics Inc. by phone at (718) 576-3205. No offer to sell any securities, and no solicitation of an offer to buy any securities, is being made in any jurisdiction in which such offer, sale, or solicitation would not be permitted by applicable law.



 Oops! We couldn’t find any results...
Oops! We couldn’t find any results...


















